 The exact split depends on the energy of the incident neutrons and the parent atoms, but generally speaking the fission products will lie in two groups, one with atomic mass in the 90-100 range and the other group in the 110-140 range. If you add up the mass of the fission products and the mass of the neutrons, alpha beta particles and neutrons, add in the kinetic energy of those plus the mas equivalence of the energy released in gamma radiation and heat etc you should get back to the original uranium mass (plus or minus some picky points). The fission products are the two, almost equal mass atoms formed by the spitting of the initiating nucleus. If you ask what the fission products of uranium are the answer will be a bit more dramatic. Also what was described in the link provided was what happens when a uranium atom absorbs a slow moving (thermal) neutron and gets a bit more energy, so it can then in turn decay into Np and Pu. Hence if you ask what Uranium is decaying into, you will get the appropriate uranium isotope decay chain, which is a sequence of small steps. I think the confusion may arise from the term ‘decay’ĭecay is normally used to describe radioactive decay where by an unstable isotope tries to loose energy by giving off alpha beta gamma radiation and all permutations of. 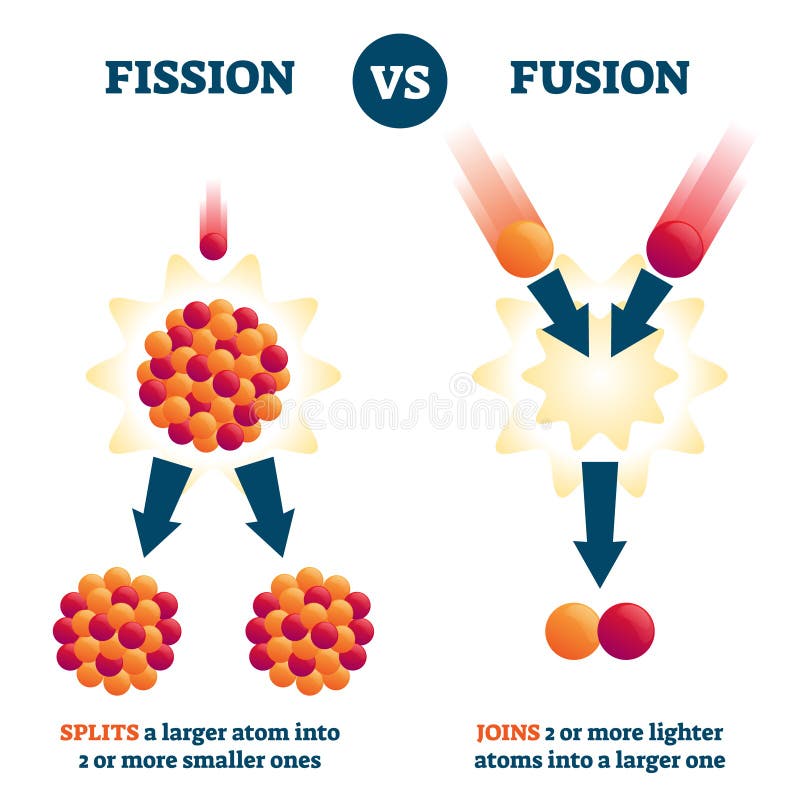
So Np and Pu? I was expecting something more radical (but mostly because I don’t know what I am talking about). I am not a physicist, but I hope that helps… That effect is used to augment the energy released by modern thermonuclear weapons, by jacketing the weapon with 238U to react with neutrons released by nuclear fusion at the center of the device. Bombarding 238U with fast neutrons induces fissions, releasing energy as long as the external neutron source is present. Instead, bombarding 238U with slow neutrons causes it to absorb them (becoming 239U) and decay by beta emission to 239Np which then decays again by the same process to 239Pu that process is used to manufacture 239Pu in breeder reactors, but does not contribute to a neutron chain reaction.įissionable, non-fissile isotopes can be used as fission energy source even without a chain reaction. But too few of the neutrons produced by 238U fission are energetic enough to induce further fissions in 238U, so no chain reaction is possible with this isotope. For example, 238U, the most abundant form of uranium, is fissionable but not fissile: it undergoes induced fission when impacted by an energetic neutron with over 1 MeV of kinetic energy. Not all fissionable isotopes can sustain a chain reaction. The second gives you the proportion of the theoretical yield that will actually result from real-world conditions. The yield of an atomic weapon can be calculated after the fact, and estimated beforehand, by taking both these into account: the first gives you the ideal theoretical yield in ergs, which can be expressed in joules or in the equivalent of kilo- or megatons of TNT. In theory, the oxidizing of carbon produces CO2 and a bunch of heat in practice, it produces CO2, heat, and some leftover carbon as soot Likewise there is an efficiency rate that is not 100% in an atomic weapon – some proportion of the uranium will not fission, but just be scattered to hellangone by the explosion resulting from the greater proportion that does. 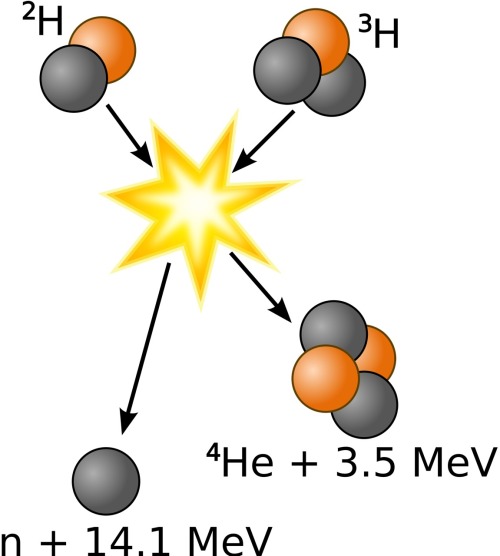 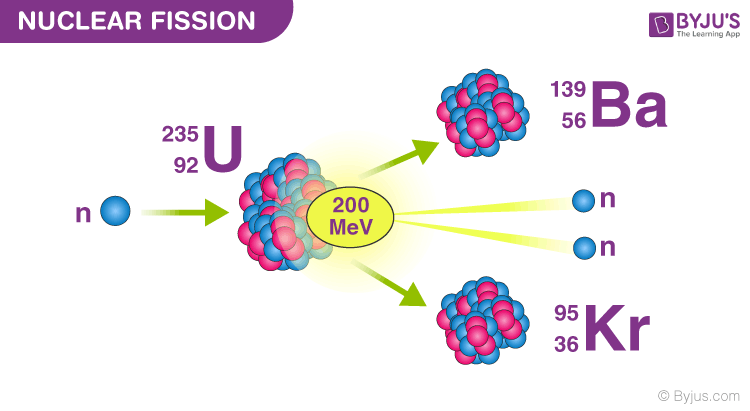
The real-world results of a theoretical thought experiment are never ideal. In fission, some quantity A of uranium is converted to several other quantities B+C+D+E of breakdown fission products, the actual weights of B, C, D, and E approaching but not quite equalling A and the (rather miniscule in mass) difference being what is converted to energy – a LOT of energy, due to the E=mc^2 equivalence. Nuclear fusion can release more energy than nuclear fission, especially when fusing small nuclei like hydrogen and helium into bigger nuclei.Only a small fraction of the mass of the warhead is converted to energy. Fission of elements heavier than iron may release energy to generate nuclei with greater binding energy (per nucleon).Fusion of elements lighter than iron may release energy to generate nuclei with greater binding energy (per nucleon).Elements with mass numbers around 60 will also be stable elements, while elements with extremely large atomic masses will be unstable. So nuclei with a mass number of approximately 60 will be the most stable, which explains why iron is the most stable element in the universe. The driving force behind fission and fusion is for an atomic nuclei to become more stable. 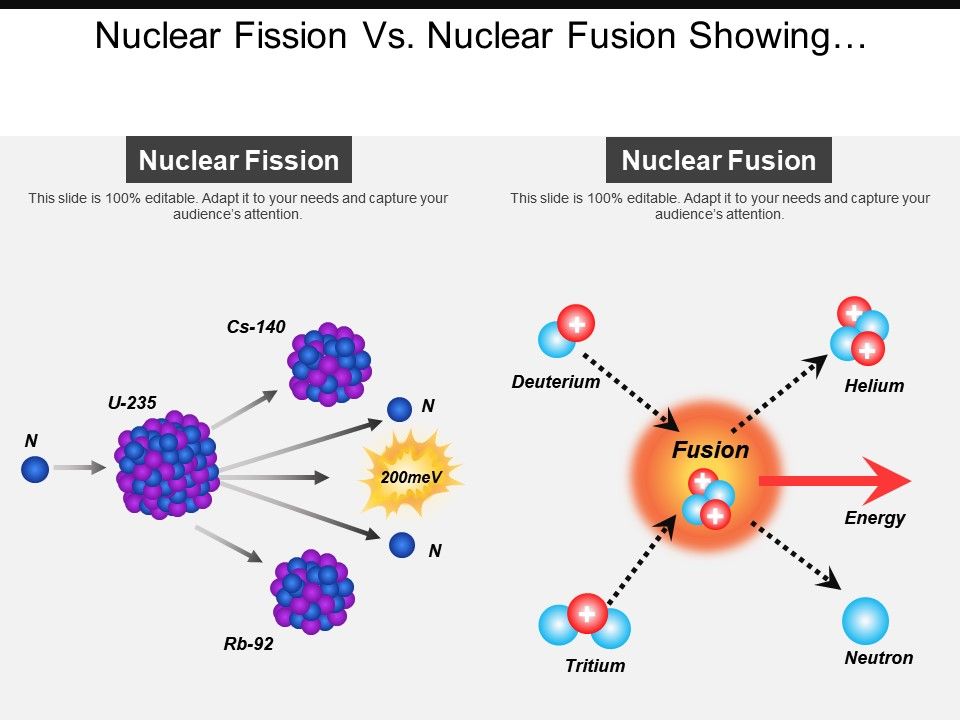
Fission processes also release energy when heavy nuclei decompose into lighter nuclei. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
